10:45 A – 12:30 A
(8:45-10:45 workout)
embryo section labeling
- quick check after 10 min hot wash in incubator at 70C in SSCT, found no sections. DAPI staining sections. Try again.
- weak BXC detectable:
- Prepped for STORM
- TRY 2: prepping new cover slip
- see if downstairs small waterbath will reach 92C for denature.
- set upstairs large incubator to 60C for pre-staining hot-pre-hybe.
- Primary only (no secondaries, see if we get less background? (Though I think this has to do with the cryo-sections, not the probe).
- denatured on hot block, at 42C O/N.
STORM
- Finished running O/N S3 Cells. Switching still looks good on final sections. 26 fields imaged (should get ~26-60 loci)
- Attempting STORM of embryo sections.
- New STORM buffers: new aliquot of Glox. New BME from Graham’s supply. The ‘new’ stock bottle is now nearly 3 years old.
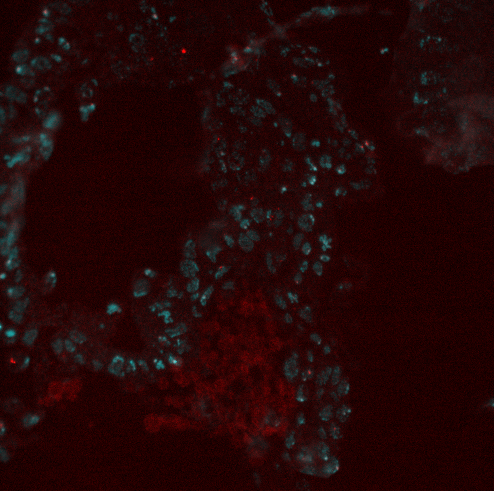
- Removed excitation filter so that DAPI cube works with 405 laser. Excellent embryo ID at 10x and 4x. Excellent embryo tiling / morphology at 100x.
- Sections look pretty good. Multiple embryos have sufficient tissue for an anterior/posterior ID and some age approximation (still not nearly as good as with resin-sectioned embryos, but maybe with more cutting practice).
- Dots look not too strong put reasonable contrast in conventional image — don’t give good STORM movies at all. Bleach out quite quickly.
- WRONG FILER SET. Samples switching just fine. Now I have 3 samples mounted to image before the buffer goes bad.
- Started re-tiling BX-C in embryos (I rather want this data set).
- Now switching BEAUTIFULLY.
- Tried to find good sections with clear anterior posterior. Can not relocate. The best set of embryos is too close to the epoxy-edge to be viewed under 100x.
- set up position list of different embryos to take O/N (at least get a decent data set of ’embryo’ BX-Cs, some range of ages and tissues. Maybe we can sort into classes.
- Set up Fab7 on STORM3.
coding
- DaoSTORM ‘3d’ option returned all molecules at position 0. Attempting ‘Z’. This does 3D fits properly.