(finally recovering from flu)
11:00a – 9:00p
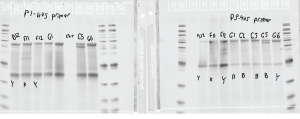
Probe making
- run denaturing gel
- G5 looks weird – what’s with the double band?! maybe give this a test anyway.
- primers are different lengths, but that doesn’t explain why both primer sets have an additional lower band that doesn’t match the other probes. Major product sizes all look correct.
- concentrations all in the 1100 ng/uL to 1500 ng/uL range
Cell staining
- RNase treating cells
- G5-P1 + G6-P3
- G1-P1 + G2-P3
- F12-P1 + G1-P3
- pre-hybing at 47C
Troubleshooting staining and beads
- EDAC treatment does seem to reduce staining. Especially long treatment.
- EDAC treatment (at least on recent samples) does not seem to keep beads attached through the 16 hr hybridization.
- Maybe EDAC has gone bad? Too much freeze thaw, too long hydrated? Ordered fresh EDAC from invitrogen early last week (12/4, not placed until 12/9, not here yet). Try crosslinking beads to cells post fixation prior to staining and see if it doesn’t interfere with staining then and if beads remain stuck.
Formaldehyde cross-linking reading
- quick overview:
- Formaldehyde becomes methylene glycol in water, two hydroxyl groups
- spectroscopic analysis of reaction of Gelatin with formaldehyde (Salsa et al 1996:) PC#1 saturates around 20-30 minutes.
first appearance of Lys-MeOH (PC#3), followed
by Arg-MeOH (PC#2) and by the Arg-Lys crosslink
(PC#l).
Ph Project
- Ajaz bringing new stained cells
- Ph-647, Flag-cy7, for M and Wt
- need to send figures
Wu lab collaboration
- need to send figures to BB
Project 2
- see notes
