9:45 am – 11:00 pm
Goals
- XZ meeting prep
- updated plots of y-internal and b-internal
- multicolor update
- low photons
- sequential method I
- sequential shadows
- toe hold probes
- Sequential staining
- test S3 first stain
- test wash out with toehold S3toe5
- test restain with S1
- Fig prep / data analysis
- crop y-internal regions for fig
- comments on manuscript (see email)
Sequential Staining
- heating objective
- heating wash buffer
- adhere beads to sample
- make new STORM buffer
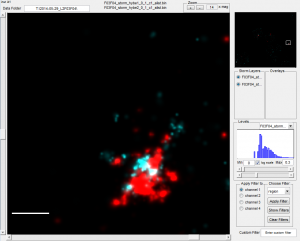
- Imaged ~8 regions of E09-P3
- Piezo not on — some notable drift may have happened 🙁
- attempting to remove S3 probe with S3toe5 at 2:1000 hybe dil buffer for 30 min at 37C (imaged cells during incubation)
- most of the decrease in signal appears to be due to bleaching, judging by near-by non-imaged regions.
- attempting to remove S3 probe with S3toe5 at 10:1000 in hybe dil buffer for 30 min at 37C
- second treatment at 1:100 dilution works! Took some images.
- washout with 50% formamide 2x SSCT at 37C ~10min
- hybe S1 at 2:1000 hybe dil buffer at 37C
- washout stain, 20 min
- add new STORM buffer and image. Dots are nice and bright
- Dots are perfectly clear in previously imaged regions, but a bit dimmer. I think the high (330 mW 647 laser) may not be ideal.
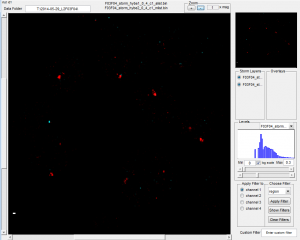
- spots not imaged don’t actually STORM that much better (imaged after rnd 2 imaging completed).
- maybe just a longer hybe and higher secondary concentration would be better?
Analysis of Sequential Stain data
- L2F03F04 from 2014-05-29
- hybe2 mostly failed (due to laser damage?)
- some images have weak staining
PH-project
- treating 1 PH-M 1 PH-Wt and 2 S2 coverslips
- 30 min in 5% FA + 1/2x PBT for post fix
- PBS wash
- 30 min in PBS + RNase A
- wash in 2X SSCT
- prehybe in 50% formamide + 2X SSCT
- stain PH-M, PH-Wt, and S2 with 750-BXC
- stain second S2 with 750 L3E09
Data analysis
- Merging old and new BXC data
- be careful, need to change source-folder for internal scaling data for BX-C back to old